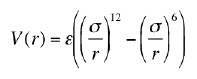|
Problems for
Intermediate Methods in Theoretical Physics
Edward F. Redish
|
The Lennard-Jones Potential
The potential energy
of interaction between two non-bonding atoms is sometimes modeled by
the Lenard-Jones (also known as the "6-12") potential:
- Sketch a qualitative graph of the L-J potential and explain what features of the curve each term is responsible for. What do these terms mean physically?
- The equation for V is sometimes written V(r) = (A/r12) - (B/r6). Discuss why one form or the other might be more advantageous.
- Find the location of the minimum of the potential, r0.
Before doing the calculation, discuss whether you expect this distance to be
greater than, equal to, or smaller than the distance σ. (Hint: Refer
to your graph in part (a).)
- Find the location of the maximum of the force between the atoms, rF0. Before doing the calculation, discuss whether you expect this distance to be greater than, equal to, or smaller than the distance you found in the previous part. (Hint: Refer to your graph in part (a).)
This function is messy to deal with. For small displacements around the minimum it's often useful to approximate the L-J potential by a shifted harmonic oscillator potential
Discuss, using a graph of the original and approximate functions, what we hope to accomplish by choosing to do this. To get the best approximation in the neighborhood of the minimum of the potential, what should we choose for V0, k and r0?
This page prepared by
Edward F. Redish
Department of Physics
University of Maryland
College Park, MD 20742
Phone: (301) 405-6120
Email: redish@umd.edu
Last revision 6. September, 2004.